Risk assessment, as defined by the National Academy of Sciences (NAS), is a systematic approach to organizing and analyzing scientific knowledge and information for potentially hazardous activities or for substances that might pose risks under specified conditions.50, 51 NAS describes the risk assessment paradigm as a process consisting of four major components: hazard identification, dose-response assessment, exposure assessment, and risk characterization. These components are described in more detail below. While the original NAS definition and paradigm reflect the risk assessment framework used today, risk assessment methodology has evolved to include new methods to reduce uncertainties and increase confidence in quantitative analyses.
It is important to recognize that risk assessment is not a single, fixed method of analysis. Risk assessment is an iterative process that involves identifying and filling data gaps in order to develop a more refined assessment of the risk. 52
The National Research Council proposed a three-phase system to ensure risk assessments are comprehensive and connected to the problems/decisions identified to render the best set of risk management options: Phase I: Enhanced Problem Formulation and Scoping; Phase II: Planning and Assessment; and Phase III: Risk Management. Phase I identifies risk management options, Phase II risks are determined using risk-assessment tools, and Phase III information gathered is used to inform risk management decisions.53
Risk Assessment (Phase II), informs the Risk Management Process (Phase III), which integrates public health, political, social, economic, engineering, and other considerations into response decisions. The relationship between risk assessment and risk management is illustrated in the diagram below, first developed by the National Research Council (NRC) in 199454 (see Figure 5).
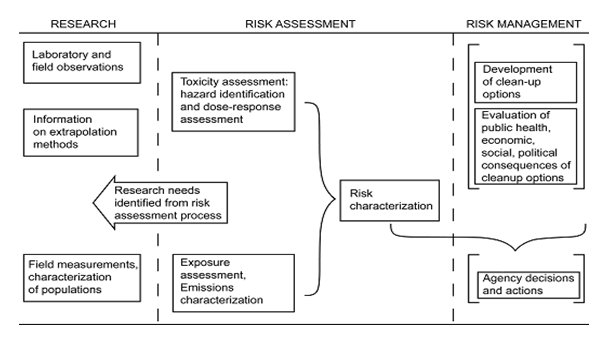
Prior to conducting a risk assessment, environmental data must be collected and analyzed. This requires planning and scoping to determine sampling and analytical needs. However, since characterization of an impacted area is an iterative process, these needs may be revisited as more information is obtained. The DHS Chemical and Security Analysis Center (CSAC) has the capacity to run risk and consequence modeling available to assist U.S. planning and response organizations.
The four components of risk assessment are described below, with hazard identification and dose- response assessment combined under the heading of toxicity assessment.
4.2.1. TOXICITY ASSESSMENT
Toxicity assessment integrates information from the hazard identification and dose-response assessment components of risk assessment.
4.2.1.1 Hazard Identification
Hazard identification is the process of determining whether an adverse health effect is likely to occur in humans and whether exposure to a particular chemical can cause an increase in the incidence of an adverse health effect in the near or long term (e.g., kidney failure, birth defects, cancer). Hazard identification involves characterizing the nature and strength of the evidence of causation.55
4.2.1.2 Dose-Response Assessment
The purpose of the dose-response assessment is to determine the relationship between the magnitude of exposure (may be expressed as an environmental concentration or internal dose) to a substance and the resultant changes in body functions (response) or health. From this quantitative dose-response relationship, toxicity values are derived that can be used to estimate the incidence of adverse effects occurring in humans at different exposure levels. Chemical risk assessments have been conducted for some chemicals and the resulting toxicity values are available in published documents. Dose-response data are often used to derive these values. Chemicals may elicit different effects depending on the exposure route (oral, dermal, or inhalation), duration, and exposure concentration. Therefore, an appropriate evaluation of the dose-response relationship should consider the duration and exposure concentration for all relevant routes of exposure, when such data are available.
In the risk assessment, the dose to an exposed individual or group is compared to available toxicity values to estimate the potential for adverse health effects. Personal sampling for chemical exposures is the preferred method to estimate exposures; however, because it is not always possible to measure the actual dose on individual, concentrations in the environment are often used as a proxy for human exposure.
Just as exposure concentration is directly related to the dose an individual receives, the dose is directly related to the severity of injury. Besides severe injury and death, more subtle toxic effects are also considered adverse. Examples of these more subtle toxic effects include potential short-term effects such as impaired mobility or altered rates of basic physiological processes (e.g., respiration, heart rate), and potential longer-term effects such as decreased general or reproductive health, or the potential to develop cancer later in life.
Another important concept related to dose is the exposure rate. With some hazardous chemicals, the degree of damage is not simply dependent on the total dose received, but also on the rate at which the dose is received and the duration of exposure. This is especially true for chemicals that are metabolized relatively quickly into nontoxic metabolites. The type of adverse health effect associated with a hazardous chemical, the exposure rate, the duration of exposure, and the specific areas of the body exposed combine to determine the type and severity of injury.
4.2.2. EXPOSURE ASSESSMENT
The objective of the exposure assessment is to estimate the magnitude of actual or potential human exposures, the frequency and duration of these exposures, and the pathways by which humans may be exposed. Conducting an exposure assessment involves: 1) analyzing contaminant releases; 2) identifying exposed populations; 3) identifying all potential pathways of exposure; 4) estimating environmental concentrations for specific pathways; and 5) estimating contaminant intakes (i.e., doses) for each pathway. An exposure pathway is the course that a chemical takes from a source to an individual. Each exposure pathway includes a source or release from a source, an environmental concentration at the point of exposure, and an exposure route. If the point of exposure is some distance from the source, a transport medium (e.g., air) is also included. The exposure route is the way the individual encounters the chemical (i.e., through inhalation, ingestion and/or dermal contact). The route is important because the toxic effects of certain chemicals vary with different routes of exposure. For example, hydrofluoric acid can cause skin burns with dermal exposures and lung damage with inhalation exposure.
Physical and chemical properties influence the likelihood of human exposure. For example, volatile chemicals or gases that are readily dispersed can quickly affect relatively large areas and have the potential to impact a greater number of people. If a nonvolatile hazardous chemical is easily dispersible or readily forms an aerosol, it poses a risk of inhalation exposure in addition to the potential for direct contact exposure. Thus, managers need to be aware that measures taken to reduce inhalation exposures may not fully address the risks of exposure via other routes such as dermal contact or ingestion.
The distribution or pattern of hazardous chemical contamination in the impacted area is a crucial variable for exposure assessment. A sampling plan is executed to define the distribution of hazardous chemicals. If the distribution is understood, then the information can be used in risk management decision-making. Even though the distribution of a hazardous chemical is necessary information to understand the potential for exposure, such information does not constitute exposure assessment by itself; it is also necessary to identify the potentially exposed populations and characterize the frequency and duration of their exposures.
The mass of the chemical in the environment and the identification of the materials and surfaces that are contaminated will assist in estimating the magnitude of the problem and the potential routes of exposure. Unfortunately, it is possible that not all hazardous chemicals will be detectable in environmental samples. For example, some chemicals may degrade in the sample container so quickly that they are no longer present in a sample by the time the analysis is performed. Alternately, their presence might be masked by other environmental contaminants, or the methods for detection might not be sensitive enough to accurately quantify the chemical. The inability to detect a particular chemical that is known to have been released should not be interpreted as the absence of the chemical. Other sources of information, including epidemiologic and forensic evidence, should be evaluated in the context of what is known about the toxicant and specific nature of the incident in question to form a hypothesis of the distribution and intensity of contamination. Such information can then be used to inform the exposure assessment.
4.2.3. RISK CHARACTERIZATION
Risk characterization combines the information about toxicity and exposures to estimate the risk for developing adverse health effects. Risk characterization also serves as the bridge between risk assessment and risk management. Major assumptions, scientific judgments, and to the extent possible, the uncertainties associated with the risk assessment and the degree to which risks may be under- or over-estimated are discussed and communicated to the decision-makers and the public.56 All information that will help inform the risk management decisions (including uncertainties and ranges for exposure and/or effect data) should be communicated clearly.
Risk assessments can be initiated at different phases of the response and can be tailored to quantify and evaluate risk to different groups for different purposes. Risk assessments for workers will incorporate regulatory occupational standards enforced by OSHA and worker focused guidelines for protective measures that are different than the standards and guidelines for protective measures used in the general population risk assessment. Although detailed, site-specific quantitative estimates of risk can be derived using data gathered during the response, qualitative risk assessments can also be developed through comparisons of measured environmental chemical concentrations to benchmarks of toxicity and exposure that have been developed by a variety of federal and state agencies: pre-calculated, health- based exposure guidelines (e.g., Acute Exposure Guideline Levels or AEGLs for short-term exposures; Regional Screening Levels for longer-term exposures; or Occupational Exposure Limits [OELs] and Immediately Dangerous to Life and Health [IDLH] values for occupational exposures). These health-based exposure guidelines are derived using equations that combine a toxicity value, a level of risk, and a set of exposure assumptions for a particular chemical, medium, and exposure scenario. Thus, the resulting health-based exposure guideline will be specific to a particular population and exposure scenario (Figure 6). For example, there are health-based exposure guidelines developed for workers that assume exposures lasting only 15 minutes and other exposure guidelines for exposures lasting 8 or 10 hours per day, 40 hours per week for a working lifetime. Likewise, there are exposure guidelines that are based on long-term/lifetime exposures to the general population that are meant to be protective of sensitive members of the population, such as children and the elderly. It should be recognized that all these approaches incorporate some degree of uncertainty in the estimated value. See Appendix A for more information.
In addition to the variables associated with the populations of concern, the exposure concentrations and the environmental persistence of the chemical contaminant may affect the magnitude of health risk associated with the exposure. One of the critical questions to be asked in performing a risk assessment associated with chemical remediation/cleanup is: “Will the hazardous chemical persist in the environment and pose a potential long-term health hazard?” The answer to this question will determine the duration of the potential exposure and the complexity and scope of the overall remediation operation. Therefore, integrating accurate information regarding persistence, total dose, toxicity, and exposure is critical to the formulation of a scientifically sound risk characterization and resultant remediation plan.
Although it may be preferred that technical staff supporting the IC develop scenario-specific remediation goals that include site- and situation-specific descriptors of exposure, in the absence of resources and site-specific information, pre-calculated, health-based exposure guidelines can provide a useful tool for risk assessors and decision-makers. However, it is important to clearly understand the basis for each exposure guideline to ensure that they are used appropriately in the response action. An overview and description of health-based exposure guidelines are presented in Appendix A.
EPA’s Risk Assessment Forum has identified four distinct time intervals that can be used to determine appropriate levels of concern for toxicity from chemical exposures.57 These include exposures for acute (<24 hours), short-term (1 to 30 days), long-term (30 days to several years), and chronic (up to a lifetime of repeated exposure) durations. Acute exposure guidelines are often prescribed for use during emergency response decisions such as evacuation/sheltering-in-place, or for emergency drinking water guidance. In modeling and some planning activities, the lowest of the acute (one-time single exposure) exposure guidelines have sometimes been used to demarcate the edge of a hazard area.58, 59 That is, these values delineate the level below which there is little to no immediate hazard to first responders for acute exposure durations. These acute exposure duration guidelines can also be used to inform decision- making regarding potential exposures to the general population during evacuation. Based on this approach, it is assumed that locations with exposure concentrations below the acute exposure guideline are less hazardous compared to those areas with concentrations above the exposure guideline.60
Chronic or long-term exposure guidelines, which are based on long-term/lifetime exposures, reside at the other end of the exposure spectrum from the acute values.61 Chronic exposure guidelines can be used as environmental screening levels or cleanup goals to evaluate chemical concentrations in different materials and surfaces and can assist in decisions regarding the extent of contamination or as a starting point for developing the ultimate clearance decision. In addition, a variety of risk assessment methods can be employed in developing risk-based, chemical-specific, site-specific radiation goals that can be used in conjunction with other site- and situation-specific information for making determinations concerning decontamination/remediation options.

Recommendations for the exposure guidelines that are most appropriate for any given situation should take into consideration the complexities and uncertainties of these determinations in order to use the available exposure guidelines most appropriately. Additionally, exposure guidelines can also be used to evaluate the adequacy of the detection limits of field- or laboratory-based analytical methods used to determine the extent and magnitude of contamination. Ideally, the full range of existing exposure guidelines should be evaluated in the context with the exposure range for the site-specific information (population exposed, duration of exposure, etc.), underlying assumptions, and other factors described in this section before determining the final cleanup goal(s).
In terms of available exposure guidelines, a wide array of quantitatively derived human toxicity and health-based exposure limits and guidelines exist for many substances. However, for certain chemicals
and certain types of environmental media, there simply may not be an existing value. In that situation, the decision-maker can consult with subject matter experts who may consider several options.62 They can review available toxicity data from animal and human studies to determine if a human exposure value could be estimated using the same modeling procedures and principles used to develop the exposure guidelines described in Appendix A.63 Another approach would be to use structural modeling, such as Quantitative Structure Activity Relationship (QSAR), or surrogate/relative potency chemical toxicity information to derive an alternative value.64 These options may have several drawbacks and may not be practical in a large-scale incident. However, while QSAR modeling may be viewed as complex, it may yield useful risk assessment information. How it is managed is what's important.


